Start ENTRESTO® with confidence
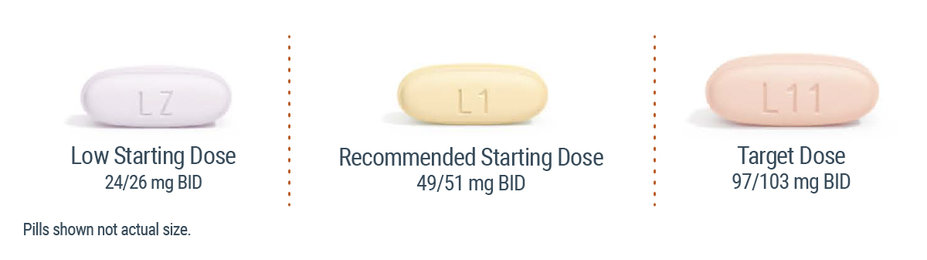
ENTRESTO film-coated tablets are available in 3 dosage strengths1
How to prescribe ENTRESTO to your adult patients
Choose initial dose of ENTRESTO based on current treatment and titrate to the target dose1
ENTRESTO is contraindicated with concomitant use of an ACEi and in patients with a history of angioedema related to previous ACEi or ARB therapy1
Avoid use of ENTRESTO with an ARB, because ENTRESTO contains the angiotensin II receptor blocker valsartan1
Unless your patient is on a high ACEi/ARB dose, start ENTRESTO at 24/26 mg twice daily and double the dose every 2 to 4 weeks, as tolerated by the patient. When switching from an ACEi, be sure to allow for a 36-hour washout period prior to initiating ENTRESTO.1 |
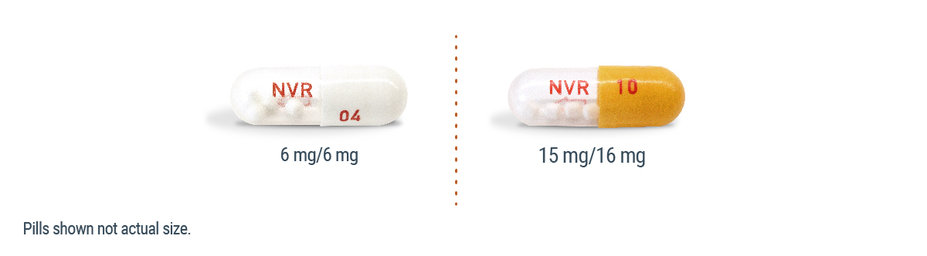
ENTRESTO SPRINKLE film-coated oral pellets within capsules are available in 2 dosage strengths1
For complete dosing and administration information, please refer to the full Prescribing Information, including Instructions for Use
*In PARAGON-HF, defined as LVEF ≥45% with structural heart disease (LAE or LVH); median LVEF was 57%. LVEF is a variable measure and the normal range can vary.1