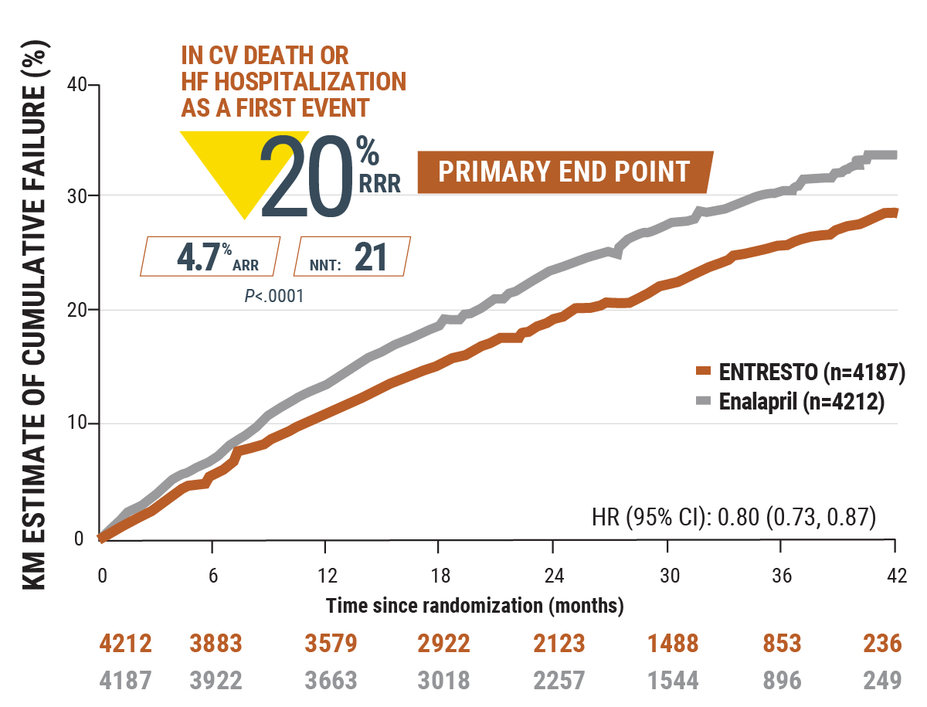
In HFrEF,
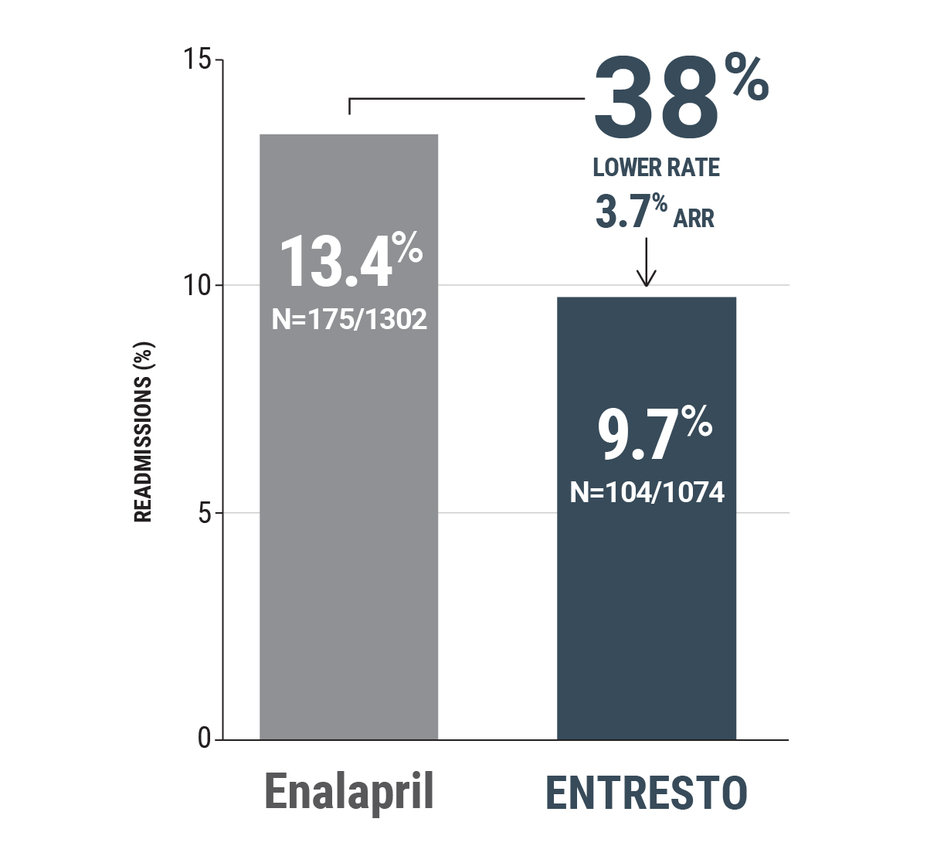
PARADIGM-HF: ENTRESTO® reduced 15-day heart failure readmissions vs enalapril
ENTRESTO demonstrated fewer 15-day heart failure readmissions in a post hoc analysis1
15-day heart failure readmissions
OR (95% CI): 0.63 (0.43–0.91)
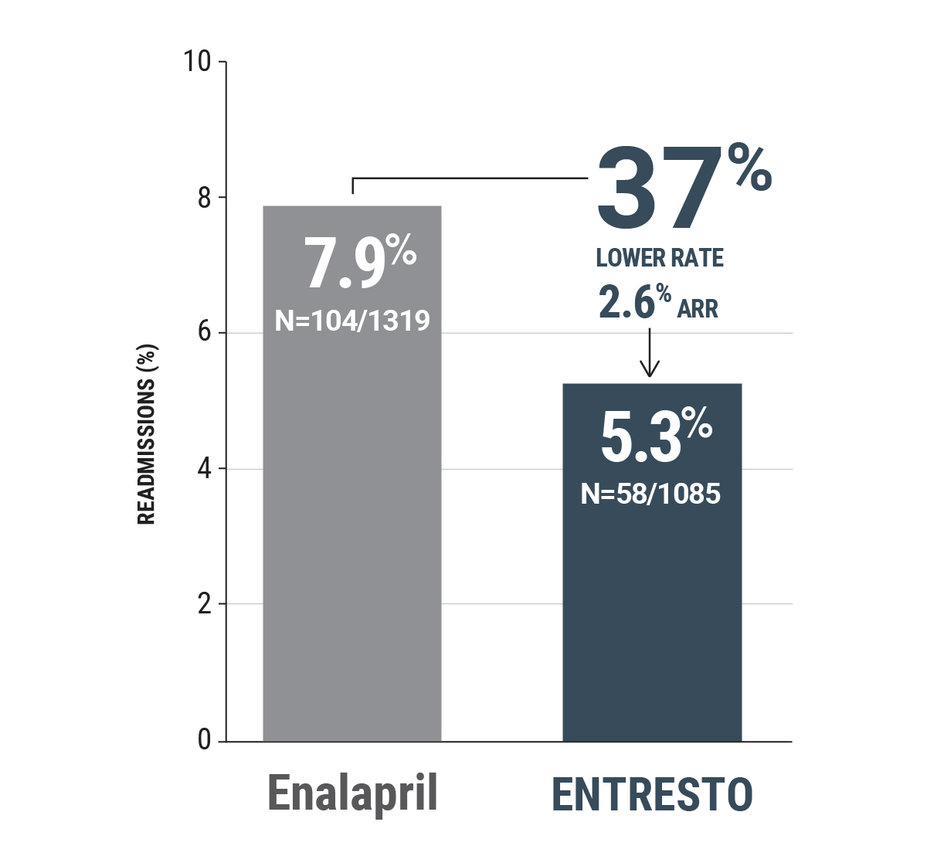
PARADIGM-HF: ENTRESTO reduced 30-day heart failure readmissions vs enalapril
ENTRESTO demonstrated fewer 30-day heart failure readmissions in a post hoc analysis1,2†‡
30-day heart failure readmissions
OR (95% CI): 0.62 (0.45–0.87)
*In PARAGON-HF, defined as LVEF ≥45% with structural heart disease (LAE or LVH); median LVEF was 57%. LVEF is a variable measure and the normal range can vary.3
PARADIGM-HF: Readmission was defined as the second hospitalization within 30 days of the first hospitalization after initiation of study drug.1
†Vs enalapril.
‡Among patients hospitalized at least once for heart failure, patient characteristics were similar at baseline between treatment groups.
PARADIGM-HF: heart failure readmissions analysis limitations2
This was a post hoc analysis of heart failure readmissions following investigator-reported heart failure hospitalizations; investigator-reported events are vulnerable to misclassification
Patients were randomized to ENTRESTO or enalapril prior to index hospitalization; the apparent differences in readmission rate could be attributed to differences in the patients
The primary unit of subsequent analysis was hospitalizations rather than patients
15-day readmissions for heart failure is not a routinely examined time point in quality and clinical assessments, and such early time points may not reflect causality
In HFrEF,
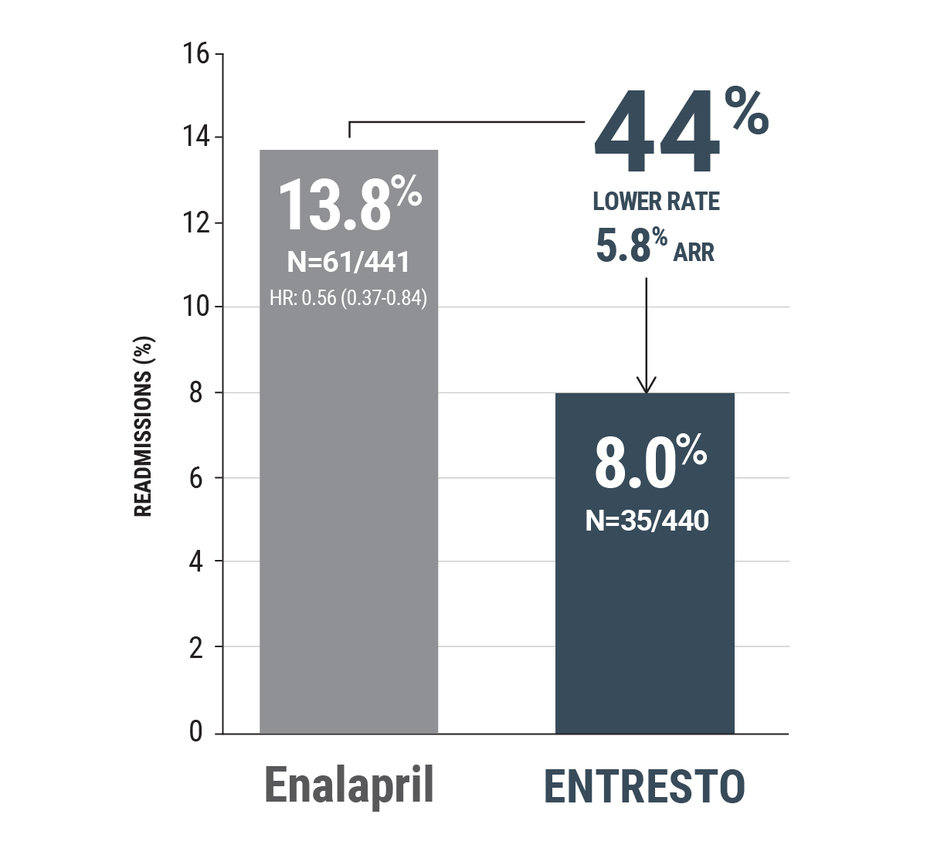
PIONEER-HF: ENTRESTO reduced 8-week heart failure readmissions vs enalapril
Inpatient initiation of ENTRESTO in stabilized patients resulted in fewer heart failure readmissions§ at 8 weeks5
8-week heart failure readmissions: prespecified exploratory end point
§PIONEER-HF: Readmission was defined as the first hospitalization after inpatient initiation of study drug.5
PIONEER-HF: study limitations5
This was a prespecified exploratory end point; the study was powered for changes in NT-proBNP
Results should be interpreted with caution due to the short time frame and infrequency of events
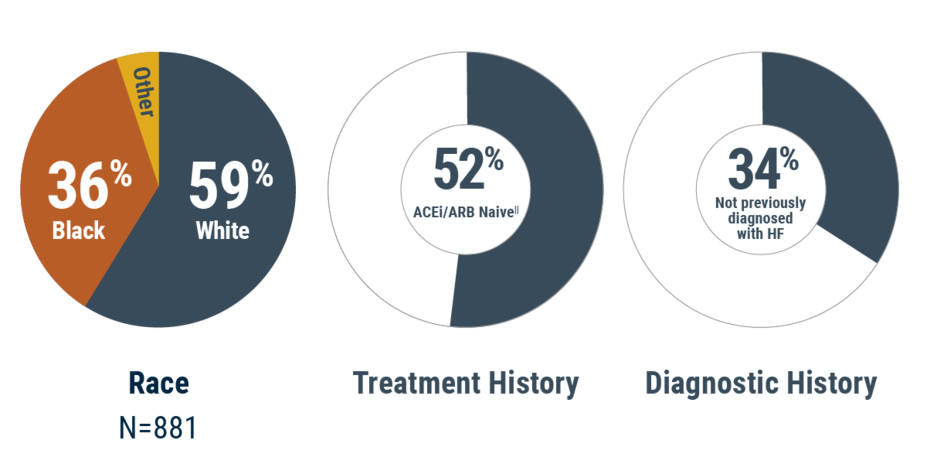
INITIATION
The median time for HFrEF patients to meet the stabilization criteria was less than 3 days after initial presentation to the hospital5
ENTRESTO was started in the hospital in a diverse set of stabilized HFrEF patients5
Patients enrolled in PIONEER-HF reflect the patients you are likely to treat in your hospital
Whether in the outpatient or inpatient setting, you can start ENTRESTO across a wide range of patient types2,3,5
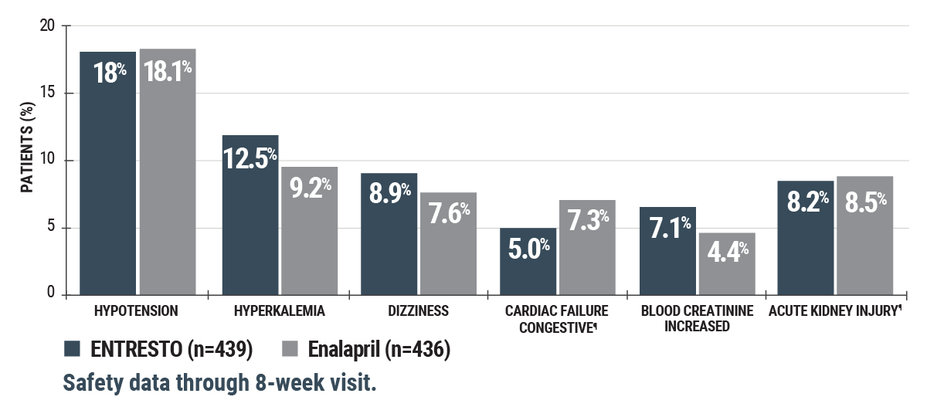
PIONEER-HF: demonstrated safety profile in HFrEF patient initiations.
¶Cardiac failure congestive and acute kidney injury were per investigator discretion on the Case Report Form.
For key safety outcomes, the ENTRESTO group and the enalapril group were generally comparable with regard to rates of worsening renal function, hyperkalemia, symptomatic hypotension, or angioedema5
Adverse event profile was comparable to that in the PARADIGM-HF trial3,5
No new safety signals were observed3,5
Safety data were collected for only 8 weeks, therefore adverse events that take longer to transpire may not have appeared in this study. Safety information should be interpreted in the context of prior trials with longer duration5