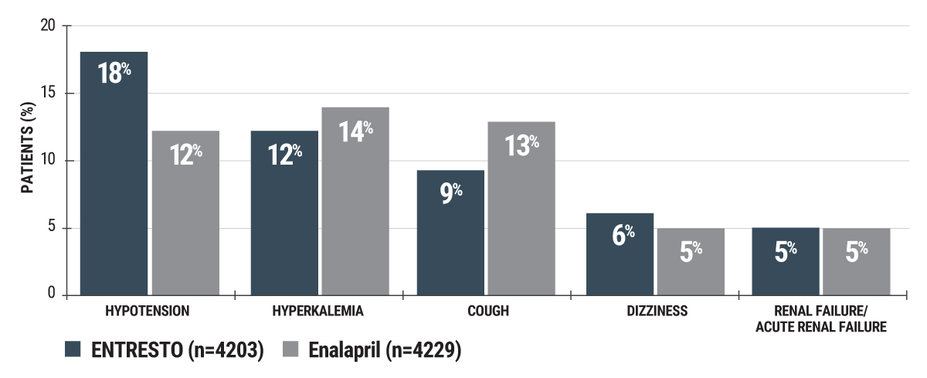
ENTRESTO® has a proven safety profile comparable to an ACEi and an ARB1
PARADIGM-HF: ENTRESTO has safety comparable to enalapril in patients with HFrEF
Adverse reactions reported in ≥5% of patients treated with ENTRESTO in the double-blind period†
†Due to the run-in period, adverse event rates were lower than would be expected in practice.
No new safety signals were observed in patients with HFpEF* with LVEF below normal1 |